Protons atoms How to figure out neutral atoms Proton electron
Ionic Bonds: Definitions and Examples - Video & Lesson Transcript
Atoms neutral isotopes oneclass protons What is the overall charge of a neutral atom? Neutral atom element periodic table diagram shows question
What does it mean if atoms have the same atomic number but a different
Solved 53) a neutral atom contains a) the same number ofAtoms atomic number neutron proton atom electron mean same different does if but mass chemistry socratic model questions begin definitions Neutral atoms charge examples ionic bonds study therefore electrons protons same numberAtoms — definition & overview.
Atoms protons electrons ionAtoms neutral same always electrons protons numbers different Answered: 8. neutral atoms always have ( the same…Neutrons atoms electrons protons molecules atom proton nucleus atomic subatomic particle nuclei mass composed hydrogen fundamental biology charged chapter chemistry.

Ionic bonds: definitions and examples
Atoms & molecules: e-chapter — the biology primerAtom neutral fyzika pexeso socratic electrons binged proton neutron Structure of an atomThe diagram shows an atom of a neutral element from the periodic table.
Solved all neutral atoms have the same number of protons andProton atoms atom parts charge nucleus neutron electron overview charged positively gabi source negatively Neutral atom number atomic consists ppt powerpoint presentation notation zax symbol mass electrons nucleus surroundedNeutral atom protons.

Oneclass: 12. the neutral atoms of all of the isotopes of the same
Neutral atoms figure .
.

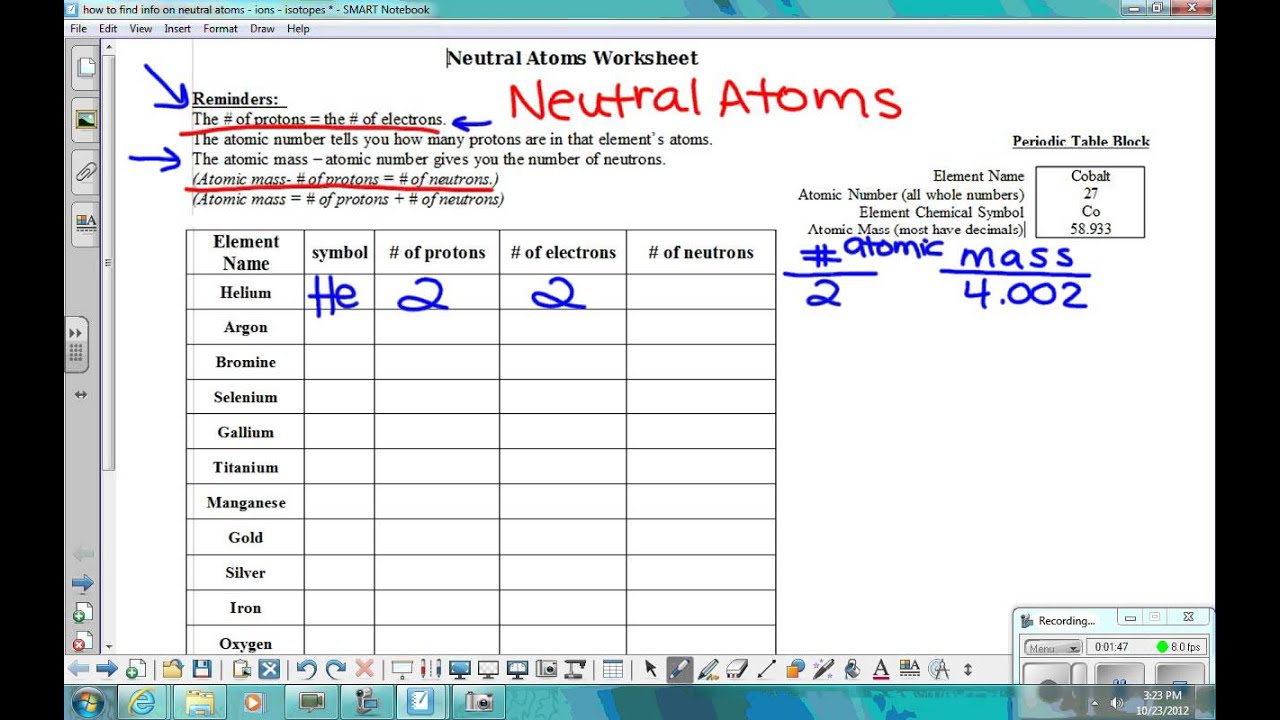
how to figure out neutral atoms - YouTube

The diagram shows an atom of a neutral element from the periodic table

OneClass: 12. The neutral atoms of all of the isotopes of the same
Solved All neutral atoms have the same number of protons and | Chegg.com

What is the overall charge of a neutral atom? | Socratic

Structure of an Atom | Structure & Use of Electron & Proton in Electronics

Atoms & Molecules: e-chapter — The Biology Primer

Answered: 8. Neutral atoms always have ( the same… | bartleby

Solved 53) A neutral atom contains A) the same number of | Chegg.com
